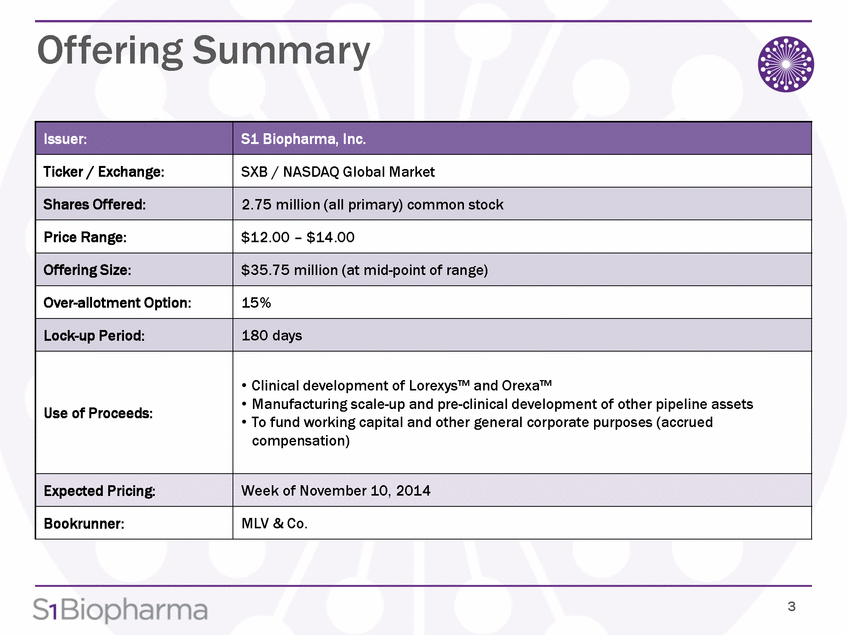
Three new buprenorphine/naloxone medications approved by FDA - Opiate Addiction & Treatment Resource

Zubsolv (Buprenorphine and Naloxone) - Maintenance Treatment for Opioid Dependence - Clinical Trials Arena
Dutch life science company Orexa secures €1.2M to develop drug to increase food intake in patients | Silicon Canals

Orexo announces last patient enrolled in pivotal study evaluating the efficacy of MODIA® in combination with sublingual buprenorphine/ naloxone for the treatment of OUD